Leukotrienes activate the immune system after infection, injury, or contact with allergens. Although their role in inflammatory processes is beneficial to help fight diseases, higher levels of these chemicals can contribute to conditions like asthma, arthritis and allergic reactions. Read on to learn more about leukotrienes and their function.
What Are Leukotrienes?
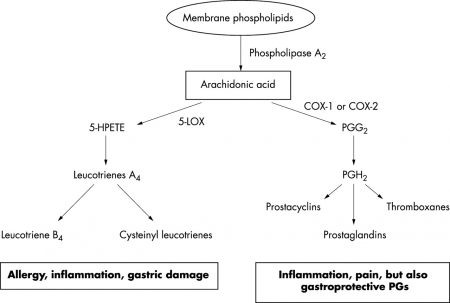
Leukotrienes are chemical messengers that powerfully activate the immune response. These molecules are formed from the breakdown of arachidonic acid (AA), a fatty acid component of cell membranes that acts as a precursor for a variety of inflammatory chemicals.
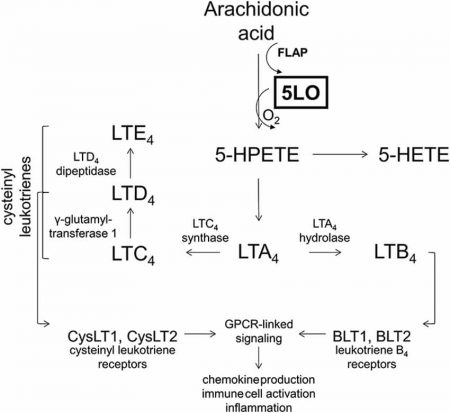
More specifically, the enzyme 5-lipoxygenase (5-LOX) converts arachidonic acid into leukotriene A4 (LTA4) in white blood cells.
Depending on the enzymes present in the cell, LTA4 is then converted into two main types of leukotrienes:
- Leukotriene B4 (LTB4): This type is formed in immune cells with the enzyme LTA4H (neutrophils and monocytes). Its main function is to promote inflammation by activating the production of inflammatory cells (neutrophils) and molecules (cytokines), and recruiting the cells to specific tissues [1].
- Cysteinyl leukotrienes (CysLT): In immune cells with the enzyme LTC4 synthase (mast cells and eosinophils), LTA4 is transformed into leukotriene C4. Outside the cells, leukotriene C4 is converted to leukotrienes D4 and E4. These types of leukotrienes tighten the airway muscles and produce excess mucus during allergic reactions [1].
Leukotriene Production
During inflammation or tissue injury, arachidonic acid is released from the cell membranes by the enzyme cPLA2 [2, 3].
The free arachidonic acid can then be broken down for the production of leukotrienes or prostaglandins [4, 5].
Source: [6]
Lipoxygenase Pathway
In the lipoxygenase pathway (LOX), 5-lipoxygenase (5-LOX) (along with other enzymes) converts arachidonic acid into two types of leukotrienes: leukotriene B4 and cysteinyl leukotrienes (comprising leukotriene C4, D4, and E4) [7, 8, 9, 10].
The 5-Lipoxygenase Pathway [11]
Functions of Leukotriene B4 (LTB4)
Leukotriene B4 has several functions during inflammation and immune defense.
1) Promotes the Migration of White Blood Cells
Leukotriene B4 recruits white blood cells (neutrophils, CD4+ T cells, and CD8+ T cells) to sites of inflammation and injury by binding to its receptor (BLT1) on these cells [12, 13, 14, 15].
In response to infections and allergic reactions, immune cells (mast cells) release leukotrienes, which attract white blood cells (neutrophils and CD8+ T cells) to the affected areas [16, 17, 18].
2) Boosts Antimicrobial Defense
In human white blood cells, leukotriene B4 stimulates the production of molecules with powerful antimicrobial effects (e.g., α-defensins) [19].
Aside from clearing away bacteria and viruses, these molecules also increase the production of leukotriene B4 [20, 21].
In mice infected with influenza A virus, leukotriene B4 significantly reduced the amount of the virus in the lung by stimulating the release of antimicrobial proteins [22].
Similarly, in a clinical trial on 23 healthy people, spray application of leukotriene B4 in the nose increased the production of myeloperoxidase (MPO) and other antimicrobial proteins after 4 hours [23].
Additionally, in cell-based studies, white blood cells (neutrophils) activated by leukotriene B4 effectively killed several viruses [23].
However, in another clinical trial study in 18 healthy people infected with the virus HRV-16, leukotriene B4 failed to reduce the incidence of common cold and its symptoms after 6 days [23].
Leukotriene B4 can also enhance phagocytosis, or the engulfment of bacteria and other disease-causing microorganisms by white blood cells (e.g., macrophages). It does this by binding to its receptor on these cells and activating cellular signals that initiate the process [24, 25].
In turn, the activated immune cells attract more cells that produce leukotriene B4, resulting in more activated immune cells [24].
Leukotriene B4 also suppresses the action of another fat messenger (PGE2) in blocking phagocytosis [26].
3) Determines the Duration of Inflammatory Responses
PPARα is a protein that promotes the production of enzymes involved in the breakdown of fatty acids and their derivatives, like leukotriene B4. Because leukotriene B4 activates PPARα, its interaction with this protein controls the inflammatory response by reducing its duration [27].
4) Activates the Immune Response
Leukotriene B4 binds to its receptor (BLT1) on dendritic cells (white blood cells that capture and digest foreign substances). This leads to the increased production of IL-12, which is required for the development of Th1 immunity [28].
Functions of Cysteinyl Leukotrienes (CysLT)
Cysteinyl leukotrienes (leukotrienes C4, D4, and E4) are mostly known for their powerful ability to constrict the airways, increase mucus production, and promote swelling and inflammation in the lungs, which worsens asthma symptoms.
1) Recruit White Blood Cells
Inhaling leukotriene E4 increased the number of white blood cells (eosinophils and neutrophils) in the mucous lining of the airways after 4 hours in patients with asthma [29].
In a cell-based study, cytokines IL-4 and IL-13 increased the production of cysteinyl leukotriene receptors (CysLT1) in human monocytes and lung macrophages (white blood cells that capture and “eat” foreign and harmful substances) [30].
Receptors for cysteinyl leukotrienes are also highly concentrated in white blood cells (eosinophils and mast cells) from nose tissues of patients with hay fever and nose inflammation [31, 32].
Similarly, the production of cysteinyl leukotriene receptors (CysLT1) is increased in white blood cells from people with aspirin-sensitive chronic nose inflammation (rhinosinusitis) [32].
2) Trigger Cytokine Production
Leukotrienes D4 and E4 trigger the release of IL-4 by eosinophils (white blood cells that fight viral and parasitic infections and cause allergic symptoms). Molecules that block cysteinyl receptors (CysLT1) prevent the production of this cytokine [33, 34].
Mast cells (white blood cells involved in allergic reactions) also release various cytokines including IL-5 and TNF-α in response to leukotriene C4 and E4 stimulation [35, 36].
3) Required for Immune Cell Transport and Function
Leukotrienes C4 and D4 restored the migration of dendritic cells (white blood cells that scavenge foreign substances) in mice lacking a protein that transports leukotriene C4 outside the cell after its synthesis [37, 38].
In asthmatic mice, cysteinyl leukotrienes triggered a Th2 response in the lungs by increasing IL-5 production from dendritic cells [39].
4) Promote Blood Vessel Leakiness
In mice deficient in either the enzyme that makes cysteinyl leukotrienes (LTC4S) or receptors for cysteinyl leukotrienes (CysLT1), blood vessel leakage was reduced by 50%. These results indicate the involvement of cysteinyl leukotrienes in increasing blood vessel leakiness [40, 41].
Other Functions of Leukotrienes
1) Enhance the Formation of White Blood Cells
By binding to cysteinyl leukotriene receptors (CysLT1) on blood and bone marrow cells, leukotriene D4 stimulates the formation of eosinophils (white blood cells involved in parasitic and allergic reactions) [42].
In mice, the addition of leukotrienes B4, C4, and D4 to bone marrow cells previously treated with blockers of leukotriene production restored the formation of various types of white blood cells [43].
Cysteinyl leukotrienes can also override the inhibition of white blood cell (eosinophil) production by other inflammatory molecules (prostaglandin E2) [44].
2) Bone Loss
Bone mass is maintained by a balance between bone formation and bone loss. During bone loss, cells called osteoclasts break down bone tissue and release its minerals, including calcium, into the blood. In cell-based studies, the addition of leukotrienes B4 and D4 to osteoclasts from birds enhanced the bone loss activity of these cells [45, 46].
Additionally, leukotriene B4 promoted the production of functional osteoclasts in human white blood cells [47].
Leukotrienes of all types were also involved in osteoclast recruitment and production during bone loss activity in mice [48].
Leukotrienes and Health
When formed excessively or inappropriately, leukotrienes can be involved in the onset or maintenance of chronic inflammatory diseases.
1) Asthma
People with asthma are very sensitive to breathing in cysteinyl leukotrienes C4, D4, and E4. These molecules narrow the airways, increase mucus production, and promote blood vessel leakiness in the lungs [49].
In addition, they recruit inflammatory cells (eosinophils) that are involved in the development of asthma into the mucus lining of the airways [50, 51, 52, 53].
Leukotriene B4 also worsens asthma symptoms. Although it does not constrict the airways, leukotriene B4 causes fluid buildup and increases mucus secretion, thereby reducing the inner width of the airways [54].
It is found in high concentration in the breath samples of patients with severe and chronic asthma [55, 56].
CD8+ T cells play a role in the development of airway constriction and inflammation. A subset of these cells containing the leukotriene B4 receptor (BLT1) has been identified in lung tissue of patients with asthma, indicating an involvement of leukotriene B4 in asthma [57].
Furthermore, in two clinical trials on 50 people, a drug (zileuton) that inhibits the enzyme that makes leukotrienes (5-LOX) improved symptoms in those with asthma [58, 59].
2) Allergies
People with hay fever (allergic rhinitis) show increased levels of all types of leukotrienes in their nose and breath [60, 61].
In addition, white blood cells from patients with asthma produce more leukotriene B4 and C4 than those from healthy individuals [62].
Furthermore, drugs that inhibit the enzyme that makes leukotrienes (5-LOX) in people with seasonal allergies reduced symptoms and leukotriene B4 levels [63, 64].
People with chronic sinus infections also respond favorably to drugs that block cysteinyl leukotriene receptors [65].
Both types of leukotrienes are involved in the development of eczema. Leukotriene B4 recruits inflammatory cells to the skin (neutrophils, eosinophils, and Th2 cells), while cysteinyl leukotrienes cause skin scarring [66].
Additionally, high levels of leukotriene B4 and C4 are found in skin lesions of people with eczema [67, 68].
In mice with eczema, scratching triggers allergic skin inflammation, which is mediated by leukotriene B4 binding to its receptor [69].
Leukotriene B4 also causes white blood cells to migrate to the mucus lining of the outer eye. This may partly explain why leukotriene B4 levels are higher in the tears of patients with seasonal allergic pink eye [70, 71].
The mucus-secreting cells of the rat and human eye contain receptors for cysteinyl leukotrienes. When stimulated by leukotriene D4, these cells secrete mucus, which is carried to the eye surface. This activity may play a role in allergic eye conditions [72].
Treatments for allergic conjunctivitis include:
- The combination of histamine H1 receptor blockers and drugs that block enzymes that make leukotriene B4 [73]
- Leukotriene B4 and cysteinyl leukotriene receptor blockers [74, 75].
In anaphylaxis, a life-threatening allergic reaction that occurs after exposure to an allergen, the increase in blood vessel leakiness is essential, as it enhances the transport of molecules promoting the reaction (leukotrienes, prostaglandins, histamine) [76].
Mice lacking the main receptors for cysteinyl leukotrienes (CysLT1) showed reduced blood vessel leakiness during skin allergic reactions. This response was increased in mice producing high levels of another type of cysteinyl leukotriene receptor (CysLT2) [41, 77].
3) Heart Disease
Leukotriene B4 is produced in plaques (build-up of cholesterol, fat, and calcium) inside the arteries [78].
In addition, patients with heart disease have higher levels of leukotriene E4 in the urine [79].
Furthermore, deficiencies in enzymes involved in leukotriene production, as well as treatment with drugs that block these enzymes, prevented artery hardening in mice [80].
The production of all the enzymes involved in the leukotriene pathway is increased in cells located in the arterial walls of patients with hardening of the arteries [81].
In mice fed a high-fat diet, deletion of the enzyme that initiates the production of leukotrienes (5-LOX) protected against an aortic aneurysm (a bulge in the wall of the aorta). However, these effects disappeared under a normal diet [82, 83].
Both the deletion of the leukotriene B4 receptor (BLT1) and treatment with a drug that blocks this receptor prevented the early development of an aneurysm in mice [84, 85].
The production of cysteinyl leukotrienes is higher in the wall of human stomach aortic aneurysms (enlargements of the stomach aorta). This causes the release of enzymes involved in the development and rupture of aneurysms [86].
Similarly, elevated levels of cysteinyl leukotrienes have been observed in both mice and human patients after brain ischemia [87, 88].
In rats, immune cells (macrophages) producing leukotriene B4 damaged blood vessels in the lungs. This effect was prevented by blocking the leukotriene B4 receptor [89].
High levels of cysteinyl leukotrienes were found in newborns with high blood pressure in the lungs. A poor clinical outcome after diagnosis was linked to high levels of leukotrienes B4, C4, and E4 [90, 91].
Leukotriene production is activated in people with aortic valve narrowing (stenosis). Its inflammatory role increases the severity of this disease [92].
4) Chronic Obstructive Pulmonary Disease (COPD)
Leukotriene B4 is higher in the exhaled breath samples of patients with COPD than in those of healthy people [93].
The recruitment of white blood cells to the airways rises during the progression of COPD and declines during its recovery [94].
5) Metabolic Disorders
Mice with type 1 diabetes exhibited higher blood leukotriene B4 levels. As a result, their white blood cells (macrophages) produced higher levels of inflammatory cytokines. Treatments with insulin or leukotriene synthesis blockers restored leukotriene B4 levels, thereby reducing inflammation [95].
Leukotriene B4 levels are elevated in fat tissue, where they trigger the production of cytokines and worsen inflammation [96, 97, 98].
The binding of leukotriene B4 to its receptor promotes insulin resistance in obese mice [99].
Furthermore, the leukotriene-producing enzyme 5-LOX is activated in both mice with insulin resistance and in fat tissue of obese patients [100].
Its deficiency reduces liver inflammation and liver cell death in mice [101].
Although its products leukotriene B4 and D4 can cause liver cell death, 5-LOX also plays a role in maintaining pancreatic function [102].
6) Cancer
Leukotriene B4 levels are increased in human colon and prostate cancer tissues [103, 104].
In addition, the production of its receptor is higher in human pancreatic tumors [105].
Also, in rats, blocking the enzyme involved in making leukotriene B4 reduces esophageal cancer [106].
In cell-based studies, high levels of leukotriene D4 led to an increased production of colon COX2, which promotes colon cancer [107].
Cysteinyl leukotriene receptors (CysLT1) are also very abundant in human colon and prostate cancer. Higher concentrations are linked to poor survival [108, 109].
Additionally, leukotrienes of both types enhance the survival, stickiness, and migration of colon cancer cells [110, 111].
However, cysteinyl leukotrienes have opposing effects in colorectal cancer. Depending on which receptor they bind to, they can promote (CysLT1) or reduce (CysLT2) cancer cell reproduction [112].
7) Rheumatoid Arthritis
Leukotriene B4 causes inflammatory white blood cells (neutrophils) to stick to the walls of blood vessels, which promotes rheumatoid arthritis development by activating the inflammatory response [113].
Additionally, the production of leukotriene B4 receptors is increased in joint tissues and cells from people with rheumatoid arthritis [114].
The severity of this disease is reduced with treatments that blocks these receptors [115].
The deletion of a protein that activates the enzyme responsible for leukotriene production (5-LOX) reduced the severity of rheumatoid arthritis by 73% and its incidence by 23% in mice [116].
Similarly, the presence of functional enzymes that make leukotriene B4 are required for mice to develop the disease [117].
8) Neurodegenerative Diseases
By activating the inflammatory NF-kB pathway, leukotriene D4 increases enzymes that produce amyloid-β, ultimately increasing the risk of Alzheimer’s disease [118].
Eliminating the enzyme required for leukotriene production (5-LOX) slows the progression of Alzheimer’s in mice [119, 120].
Interestingly, people with Alzheimer’s disease show increased production of this enzyme and leukotriene B4 levels compared to healthy individuals [121].
Similarly, in mice brain cells treated with a neurotoxin, the production of an enzyme that initiates leukotriene synthesis (5-LOX) and leukotriene B4 was increased, causing brain cell death. The addition of a blocker of this enzyme promoted the survival of these brain cells [122].
9) Stomach Pain
In people with familial Mediterranean fever (FMF), an inflammatory disorder causing stomach pain, leukotriene B4 levels in urine are higher than those of healthy people [123].
Additionally, high concentrations of leukotrienes B4, C4, and D4 are found in the stomach juices of children infected with Helicobacter pylori, thus causing stomach pain [124].
In 5 children suffering from food allergies, blocking leukotriene receptors with drugs (montelukast sodium) prevented stomach pain during a year of oral immunotherapy [125].
This strategy also relieved symptoms of stomach pain in other inflammatory diseases like Henoch-Schönlein purpura, mastocytosis, and eosinophilic gastroenteritis [126, 127, 128].
Conversely, stomach pain was the most common non-psychiatric side effect observed in children with asthma or early wheezing treated with leukotriene receptor blockers [129].
10) Pain Sensitivity
After nerve injury, the levels of leukotriene B4 and its receptor (BLT1) are higher in spinal nerve cells. This enhances the activity of receptors involved in pain (NMDA), ultimately increasing pain sensitivity [130].
Similarly, leukotriene B4 binding to its receptor enhances the activity of a receptor involved in inflammatory pain (TRPV1). While low concentrations of this leukotriene trigger this process, higher concentrations allow leukotriene B4 to bind to another receptor (BLT2), leading to an opposite effect [131].
11) Lung Tissue Scarring
In mice lacking the necessary agents for leukotriene production, chronic lung inflammation and tissue scarring were reduced [132, 133, 134].
Further Reading
Takeaway
Leukotrienes are compounds made from arachidonic acid which activate the immune and inflammatory responses. They are required for healthy immune reactions to pathogenic microbes, and they attract and activate white blood cells to prevent and resolve disease.
Excess leukotrienes may promote an unhealthy inflammatory reaction. People with asthma, allergies, heart disease, metabolic disorders, and other inflammatory conditions tend to have higher levels of leukotrienes than healthy individuals. Leukotrienes may also increase sensitivity to pain and promote bone loss.